
Episodes

Monday Sep 08, 2025
Monday Sep 08, 2025
In this episode, Dr. Aditya Joshi and coauthors discuss their team’s recently published study, “Cinnabarinic acid protects against metabolic dysfunction-associated steatohepatitis by activating Aryl hydrocarbon Receptor-dependent AMPK signaling." The study showed that absence of AhR in hepatocytes results in exacerbated metabolic dysfunction-associated steatohepatitis (MASH) in mice subjected to Western-style high-fat high-fructose high-cholesterol diet. Moreover, treatment with a tryptophan catabolite, cinnabarinic acid (CA) mitigated hallmarks of MASH in an AhR-dependent manner. In conclusion, the study delineates the significance of hepatic AhR-dependent AMPK signaling in CA-mediated protection against MASH.
Nikhil Y. Patil, Iulia Rus, Felix Ampadu, Hassan M. Abu Shukair, Sarah Bonvicino, Richard S. Brush, Elena Eaton, Martin-Paul Agbaga, Tae Gyu Oh, Jacob E. Friedman, and Aditya D. Joshi
2025 329:4, G433-G447

Thursday Jul 24, 2025
Thursday Jul 24, 2025
In this episode, coauthors Daria Igudesman and Karen D. Corbin discuss their team’s recently published study, "Microbiota encroachment and a gut-adipose-liver axis in metabolic dysfunction-associated steatotic liver disease." This first-in-human study explores how changes in the gut microbiota may contribute to metabolic dysfunction through a gut-adipose-liver axis. The researchers found that colonic expression of insulin and lipid-related genes may link microbial encroachment in the colon with adipose tissue insulin resistance and the development of metabolic dysfunction-associated steatotic liver disease (MASLD). Tune in to learn how these findings offer new insights into the complex interplay between the gut microbiome and metabolic health.
American Journal of Physiology-Gastrointestinal and Liver Physiology 2025 329:1, G201-G214

Thursday Jul 24, 2025
Thursday Jul 24, 2025
In this episode, coauthors Jazmyne L. Jackson, Abigail J. Staub, Annie D. Fuller, and Kelly A. Whelan join us to discuss their recently published review, "Mouse models of eosinophilic esophagitis: molecular and translational insights." Eosinophilic esophagitis (EoE) is a chronic allergic inflammatory disease of the esophagus with growing prevalence and clinical impact, yet many of its underlying mechanisms remain poorly understood. The authors explore how murine models have advanced our knowledge of EoE pathogenesis, highlighting key molecular and translational findings uncovered through in vivo research. This episode delves into the strengths and limitations of existing models, current challenges, and future opportunities for leveraging mouse models to guide diagnosis, monitoring, and therapy development in EoE.
American Journal of Physiology-Gastrointestinal and Liver Physiology 2025 329:1, G215-G231

Thursday Jul 24, 2025
Thursday Jul 24, 2025
In this episode, Jayson M. Antonio and Ronaldo P. Ferraris discuss their team’s recently published study, "Lacticaseibacillus rhamnosus GG-driven remodeling of arginine metabolism mitigates gut barrier dysfunction." The research reveals a novel probiotic-driven mechanism that links dietary tryptophan with host arginine metabolism. The probiotic Lacticaseibacillus rhamnosus GG, when combined with tryptophan, enhances gut barrier integrity by upregulating argininosuccinate lyase (ASL), a key enzyme in arginine biosynthesis. The study also identifies ASL downregulation and increased serum argininosuccinate levels in a mouse model of colitis, pointing to a promising target for precision probiotic therapies.
American Journal of Physiology-Gastrointestinal and Liver Physiology 2025 329:1, G162-G185

Thursday Jul 24, 2025
Thursday Jul 24, 2025
In this episode, we’re joined by coauthors Preedajit Wongkrasant and Keith A. Sharkey to discuss their recent publication, "Fructooligosaccharides slow colonic motility and activate myenteric neurons via calcium sensing and 5-HT3 receptors in the proximal colon." Calcium-sensing receptors are known to play an important role in gastrointestinal physiology. This study reveals a new function of fructooligosaccharides, a type of dietary fiber, in regulating colonic motility and activating the enteric nervous system. The effects are mediated through calcium-sensing and 5-HT3 receptors, providing new insights into gut–brain communication and potential therapeutic targets for gastrointestinal disorders.
American Journal of Physiology-Gastrointestinal and Liver Physiology 2025 328:6, G734-G745
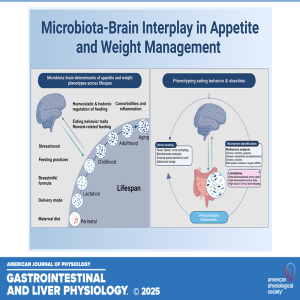
Thursday May 22, 2025
Unraveling Appetite: The Microbiota-Brain Connection Across the Lifespan
Thursday May 22, 2025
Thursday May 22, 2025
Monday Apr 14, 2025
Unraveling Fabry Disease: Lyso-Gb3’s Role in Gut Physiology
Monday Apr 14, 2025
Monday Apr 14, 2025
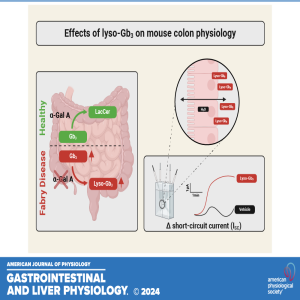
In this episode, coauthors Cecilia Delprete, Friederike Uhlig, Marco Caprini, and Niall P. Hyland dive into their latest research titled "Characterization of Fabry disease-associated lyso-Gb3 on mouse colonic ion transport and motility."
Fabry disease (FD) is a rare genetic disorder with wide-ranging effects across the body, including the gastrointestinal (GI) system—often one of the first to show symptoms. The team explores how the lipid molecule lyso-Gb3, known to accumulate in FD, affects colonic function using ex vivo techniques like Ussing chambers and organ baths.
The conversation covers key findings: lyso-Gb3 significantly alters ion transport in the colon but doesn't affect motility or nerve responses, suggesting a complex mechanism behind GI symptoms in FD patients. Could this bring us closer to understanding—and eventually treating—the GI discomfort that burdens so many with Fabry disease? Tune in today!
American Journal of Physiology-Gastrointestinal and Liver Physiology 2024 327:6, G810-G817

Thursday Mar 13, 2025
Got Guts: A Chance to Put Liver Biopsy Back to Its Former Glory?
Thursday Mar 13, 2025
Thursday Mar 13, 2025
In this episode, coauthors Camilla Venturin and Luca Fabris delve into their editorial titled "Machine Learning Application to Histology for the Study of Cholangiopathies (BiliQML): A Chance to Put Liver Biopsy Back to Its Former Glory?" The episode explores groundbreaking research by Dominick Hellen and colleagues on the limitations of current histological techniques in studying cholangiocytes and the biliary tree.
Historically, the study of these cells has been constrained by outdated and error-prone methodologies, like two-dimensional cell counting or complex three-dimensional imaging that fails to provide reliable quantification. Enter BiliQML, a novel machine learning model that promises to revolutionize this field. This episode breaks down how BiliQML quantifies biliary forms using anti-Keratin 19 antibody-stained whole slide images, providing a far more accurate and scalable approach.
With an impressive F-score of 0.87, the model's application across a variety of cholangiopathy models, including genetic, surgical, toxicological, and therapeutic, showcases its sensitivity and robustness. The episode reveals how this cutting-edge technology opens new doors for both clinical and basic-science researchers in the field of cholangiopathies. Tune in to discover how machine learning is bringing liver biopsy back to the forefront of research.
American Journal of Physiology-Gastrointestinal and Liver Physiology 2024 327:6, G733-G736
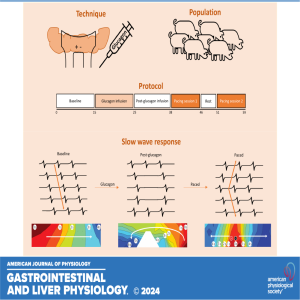
Friday Feb 21, 2025
Friday Feb 21, 2025
In this podcast coauthors Nipuni D. Nagahawatte and Leo K. Cheng discuss their research titled "High-energy pacing inhibits slow-wave dysrhythmias in the small intestine." Glucagon was infused in pigs to induce hyperglycemia and the resulting slow-wave response in the intact jejunum was defined in high resolution for the first time. Subsequently, with pacing, the glucagon-induced dysrhythmias were suppressed and spatially entrained for the first time with a success rate of 85%. The ability to suppress slow-wave dysrhythmias through pacing is promising in treating motility disorders that are associated with intestinal dysrhythmias.
American Journal of Physiology-Gastrointestinal and Liver Physiology 2024 326:6, G676-G686

Thursday Jan 23, 2025
Thursday Jan 23, 2025
